Our Service

-
At Aland Enterprise, we work with our partners to provide the best match medical import and export consultation and planning for their success. Such consultation includes, but is not limited to, regulatory research, market research and submission of regulatory certificates and notifications. Aland Enterprise LLC. does not offer any legal or medical advice to any clients or partners. Our partnering scientific staff comprises more than 90 professionals, including experts in toxicology, global regulatory affairs, chemistry, nutrition, and risk assessment analysis. together we specialize in developing scientific and strategic global regulatory plans, resolving complex toxicological and scientific issues, and facilitating timely regulatory approvals. Our experts understand the specific and unique challenges of their clients and are well-equipped to assist with these needs.
-
Item descriptionIn the United States, pet food is regulated primarily under the authority of the Food and Drug Administration (FDA) pursuant to the Federal Food, Drug, and Cosmetic Act. Pet food products must be manufactured from ingredients that are either approved food additives, Generally Recognized as Safe (GRAS), or defined and accepted through the Association of American Feed Control Officials ingredient definitions. AAFCO establishes model regulations for labeling, ingredient definitions, and nutritional adequacy statements, which are adopted and enforced at the state level. Manufacturers are responsible for ensuring that products are safe, properly labeled, and produced under sanitary conditions in compliance with Current Good Manufacturing Practices (cGMPs) under 21 CFR Part 507. If a novel ingredient is introduced, it must undergo either a food additive petition process or GRAS notification to the FDA. Additionally, state-level feed registration is required prior to distribution, and nutritional claims must be substantiated either through formulation to AAFCO nutrient profiles or feeding trials. Continuous compliance requires documentation, traceability, hazard analysis, and preventive controls under the Food Safety Modernization Act (FSMA).
-
Pet wellness supplements for dogs and cats occupy a more nuanced regulatory position and are also overseen by the Food and Drug Administration through its Center for Veterinary Medicine (CVM). These products are generally regulated as animal feed or feed supplements rather than drugs, provided that they do not make disease treatment, prevention, or cure claims. Ingredients must still be either GRAS, approved food additives, or recognized under Association of American Feed Control Officials definitions. If a substance is new or lacks established safety consensus, a GRAS determination or food additive approval is required prior to market entry. The National Research Council provides scientific reference standards for nutrient requirements, which may support formulation and safety rationale. Structure/function claims (e.g., “supports immune health”) are permitted, but therapeutic claims will trigger classification as a new animal drug requiring formal FDA approval. As with pet food, manufacturers must comply with cGMPs, maintain safety and substantiation records, and ensure accurate labeling. Ongoing regulatory compliance includes ingredient verification, adverse event monitoring, and readiness to provide documentation demonstrating safety and regulatory status upon inspection.
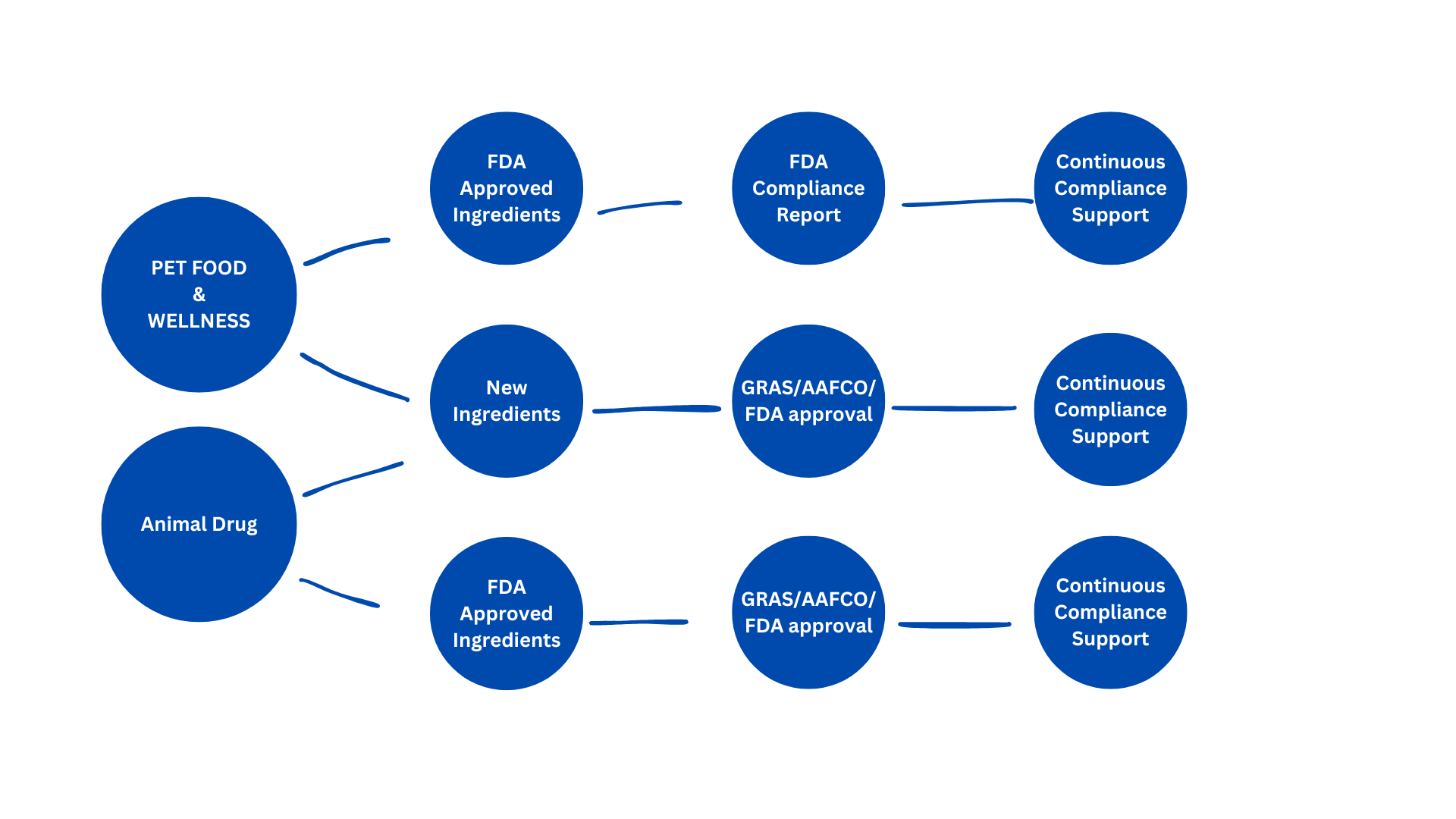
Regulatory compliance is indispensable for all animal food/wellness/drug products.
Variable cost for different products.
If it’s a new animal food, the pathway is very different from drugs—but still regulated tightly. The key issue is not the product itself, but whether each ingredient is legally allowed.
Aland Enterprise or our partners/agents/contractors do not provide legal or medical advice.
For more information, please go to
https://www.fda.gov/animal-veterinary/animal-foods-feeds/pet-food
https://www.fda.gov/animal-veterinary/animal-health-literacy/fdas-regulation-pet-food
https://www.fda.gov/animal-veterinary/products/animal-foods-feeds
https://www.fda.gov/animal-veterinary/animal-foods-feeds/animal-food-labeling-and-pet-food-claims
https://www.fda.gov/animal-veterinary/animal-foods-feeds/animal-food-ingredients
Animal Drugs Information:
Important Compliance Information:
